PPE is the most important measure to prevent the spread of infectious diseases within the medical sector and the demand is rising continuously especially due to the outbreak of COVID-19 and the presence of counterfeited products entering the market causes that some gloves on the market might be sub-standard.
BEAM Analytical provide a comprehensive range of tests on PPE especially on disposable glove testing capabilities to support both manufacturers and distributors in ensuring your disposable gloves comply with industry and performance standards.
Medical gloves are classified as Class II devices, and must be licensed as such. This is the case whether the gloves are made of latex, vinyl, synthetic polymer or nitrile, or are sterile or non-sterile. For example, disposable, non-sterile, polyethylene gloves in first aid kits are medical gloves. They protect both the patient and the wearer.
The Canadian standard is for glove to be compliant to ISO 11193-1, or to ASTM D3578 depending on their material. However, if a glove is EN 455 compliant, then it also meets the required standard.
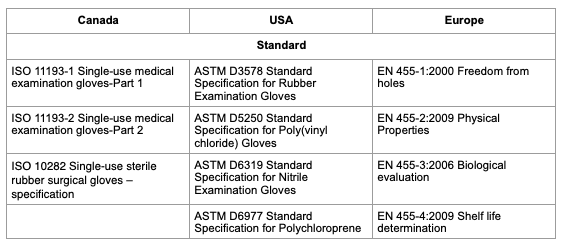
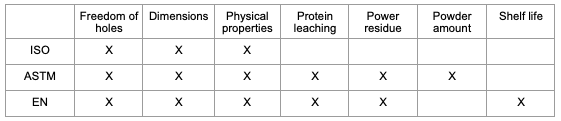
Each standard has its own specifications, but cover the following: